The overall goal of this project is to design a device that can be used in an emergency for someone undergoing anaphylaxis, mainly as a more cost-effective, safe, and efficient alternative to existing devices. Our specific goals include a cheaper device (under $300), a less invasive epinephrine administration method, accomodation for patients of different body types, and an accurate and precise delivered dose of epinephrine. To create this device, we must explore different solutions to anaphylaxis that include alternate routes of epinephrine application, safe and effective patient use, and any additional features desired by our client (Dr. Caroline Horner, WUMS) and her patients. We will aim to have a proposed design by the end of first semester and a finished design by the end of the academic year in April 2017. The device should provide a consistent but temporary treatment in the case of anaphylaxis.
Design
Our design team has determined that an Intramuscular (IM) method is superior to others, including subcutaneous, inhalable, intranasal, or other methods. Our client has specified that this would be the best option to pursue and that creating a new product using a different delivery method was inadvisable. We believe that although it would be difficult to devise a device that used an alternative drug delivery method, that is not completely out of the question for the future.
We are planning on including new feature in our IM epinephrine injector to make it the most innovative product on the market. Our design will include variable needle length, separate ingredient mixing chambers to create temperature independence, varying doses for patients of different needs, a retractable needle, and a replaceable needle-cartridge system with straight-forward instructions.
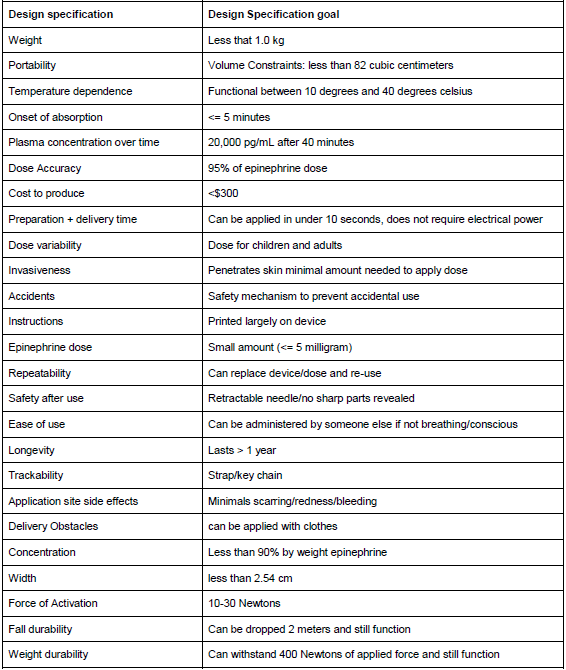
Complete Design Specifications:
We are planning on including new feature in our IM epinephrine injector to make it the most innovative product on the market. Our design will include variable needle length, separate ingredient mixing chambers to create temperature independence, varying doses for patients of different needs, a retractable needle, and a replaceable needle-cartridge system with straight-forward instructions.
Complete Design Specifications:
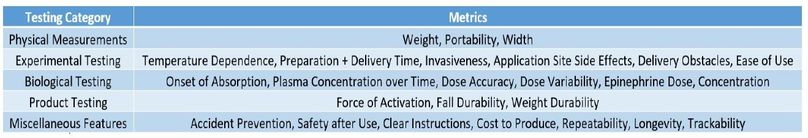
Design Verification Categories:
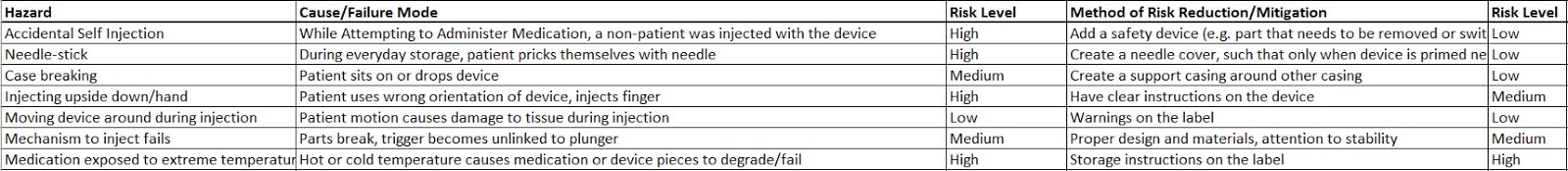
DesignSafe:
Schedule
Our design project will follow the approximate schedule below. This may change due to unforeseen circumstances.
Project Proposal: October 7 (Presentation)
Primary Concept Selection: November 15th
Preliminary Design Features and Progress Report: December 5th (Presentation)
Finalized Design: February 20th
Verification & Validation: March (Presentation)
Initial Prototype Finished: March 30th
Prototype Testing: April 7th
Final Modifications and Retesting: April 20th
Final Presentation: Late April (Presentation)
Project Proposal: October 7 (Presentation)
Primary Concept Selection: November 15th
Preliminary Design Features and Progress Report: December 5th (Presentation)
Finalized Design: February 20th
Verification & Validation: March (Presentation)
Initial Prototype Finished: March 30th
Prototype Testing: April 7th
Final Modifications and Retesting: April 20th
Final Presentation: Late April (Presentation)